Abstract:
JC. Trullas1, JL. Morales2, J. Casado3, M. Carrera4, M. Sanchez-Marteles5, A. Conde-Martel6, MF. Davila7, P. Llacer8, P. Salamanca-Bautista9, J. Perez-Silvestre10, MA. Plasin11, JM. Cerqueiro12, P. Gil13, F. Formiga14, L. Manzano15 – (1) Hospital d’Olot I Garrotxa County, Olot, Spain (2) Hospital Arnau de Vilanova, Internal Medicine Department, Lleida, Spain (3) University Hospital of Getafe, Internal Medicine Department, Getafe, Spain (4) Complejo Hospitalario de Soria, Internal Medicine Department, Soria, Spain (5) Clinical University Hospital Lozano Blesa, Internal Medicine Department, Zaragoza, Spain (6) University Hospital Dr Negrin, Internal Medicine Department, Las Palmas De Gran Canaria, Spain (7) University Hospital Nuestra Se?ora de Candelaria, Internal Medicine Department, Santa Cruz De Tenerife, Spain (8) Hospital de Manises, Internal Medicine Department, Valencia, Spain (9) Hospital Universitario Virgen Macarena, Internal Medicine Department, Sevilla, Spain (10) Hospital General Universitario de Valencia, Internal Medicine Department, Valencia, Spain (11) Mollet Hospital, Internal Medicine Department, Mollet Del Valles, Spain (12) Lucus Augusti University Hospital, Internal Medicine Department, Lugo, Spain (13) La Princesa University Hospital, Internal Medicine Department, Madrid, Spain (14) Bellvitge University Hospital, Internal Medicine Department, Hospitalet De Llobregat, Spain (15) Ramon and Cajal University Hospital, Internal Medicine Department, Madrid, Spain
Background:
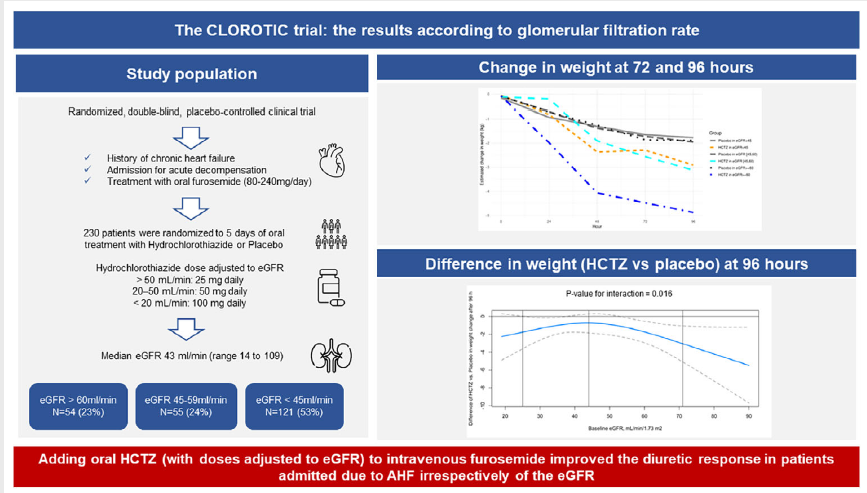
Persistent congestion is an important clinical target in acute decompensated heart failure (HF), typically addressed using loop diuretics, but an important and challenging subset of HF patients exhibit fluid overload despite significant doses of loop diuretics.
Methods:
In a prospective, double-blind, placebo-controlled, randomized trial, we assigned 230 patients with chronic HF who presented an acute decompensation to receive hydrochlorothiazide (HCTZ) or placebo in addition to a pre-specified furosemide regimen. The coprimary end points were changes in body weight and changes in patient-reported dyspnea from baseline to 72 hours of hospitalization. Secondary outcomes included changes in body weight and changes in patient-reported dyspnea from baseline to 5th day of hospitalization, metrics of diuretic response, hospital length of stay, mortality and rehospitalizations (all-cause and HF-related) at 30 and 90 days. Safety outcomes (changes in renal function and changes in electrolyte levels [sodium and potassium] were also assessed.
Results:
A total of 230 patients (48% women) were randomized. Median age was 83 years, and the mean left-ventricle ejection fraction was 55% (65% had an ejection fraction of 50% or greater). Patients assigned to the HCTZ were more likely to lose weight 72 hours after randomization than those assigned to placebo (-2.3 vs. -1.3; P=0.001) and had a non-significant trend toward greater improvement in patient assessment of symptoms (mean AUC for dyspnea at 72 hours using VAS [visual analogue scale]; 960 vs. 720, respectively; P=0.449). These results were similar at day 5 after randomization. Patients assigned to HCTZ presented a significantly greater 24-hour diuresis (1775 vs. 1400mL; P=0.037) and weight loss for each 40 mg of furosemide (both at 72 hours and at 5 days) (p<0.001) and a nonsignificant trend toward higher net fluid loss per milligram of furosemide (786 vs. 719; P=0.255). The mean loop diuretic dose administered from enrolment to 72 hours was 375 mg in the placebo group and 340 mg in the HCTZ group (P=0.145) and the proportion of patients that could reduce the dose of loop diuretic was higher in the HCTZ group in comparison with placebo (31.5% vs. 17.4%; p=0.045). There were no differences in length of stay, mortality or rehospitalizations at 30 and 90 days. A higher proportion of patients who received HCTZ met the prespecified safety end point of impaired renal function as compared with placebo (46.5% vs. 17.2%; P<0.001) but changes in serum sodium and potassium were similar between the two groups.
Conclusions:
Adding treatment with oral HCTZ to intravenous furosemide was a safe and effective strategy to improve diuretic response in acute decompensated HF.










Leave a Reply